Sludge bulking remains one of the most persistent challenges in small and mid-sized wastewater treatment facilities, particularly during seasonal shifts that affect the food-to-microorganism (F/M) ratio, nitrogen-to-phosphorus (N/P) ratio, and temperature. These operational changes often trigger the overgrowth of filamentous organisms or biofilms, leading to poor sludge settling, high sludge volume index (SVI), and inefficiencies in the secondary clarifier.
In recent years, a promising solution has emerged: reactive oxygen species (ROS), especially hydroxyl radical ions, which offer a more targeted and effective approach to breaking down biofilm structures. One ROS-based treatment, JC-9465, has demonstrated remarkable success across several small-scale treatment facilities—including a notable case in Southern California.
Field Testing and Findings
Over the past six years, JC-9465 has been evaluated in small wastewater treatment systems (less than 2 MGD) under varying seasonal and operational conditions. Fluctuations in F/M ratios, nutrient imbalances, and temperature swings consistently resulted in sludge bulking and poor settling—regardless of geographic location or process design. It was found that the underlying problem was eventually traced back to microbial imbalances and the presence of biofilms.
At one test site, dosing 25–30 mg/L of JC-9465 into the Return Activated Sludge (RAS) line yielded dramatic improvements. The treatment broke down the extra polymeric substances (EPS) in the biofilm matrix and effectively regulated microbial populations—without disrupting the biological process. Within 7–10 days, settling performance normalized, and the system recovered without resorting to super-chlorination or reseeding.
It’s important to note that traditional bulking control often focuses on removing filamentous bacteria. However, in many of these studies, the presence of filamentous organisms was either minor or absent altogether. Instead, dense biofilms, composed of EPS, were found to be the primary culprit. These EPS-laden structures hinder proper floc formation and settling by creating buoyant microenvironments that trap solids. This insight marked a shift in treatment strategy—from targeting specific microorganisms to disrupting the physical matrix of the biofilm itself.

Role of Biofilm and EPS
Microscopic monitoring during the trials revealed two dominant types of microorganisms contributing to sludge bulking: filamentous bacteria and non-filamentous floc-forming microbes. However, what proved most consistent and problematic was not the specific type of organism—but the presence of EPS-rich biofilms.
EPS (Extra Polymeric Substances) are the “glue” that holds biofilms together, composed of complex macromolecules such as polysaccharides, proteins, lipids, and nucleic acids. These substances create protective channels and structures that resist mechanical disruption and chemical penetration. Within this matrix, even healthy floc can be suspended rather than settling, leading to chronic bulking issues. Traditional flocculants and oxidants often fail to fully penetrate this structure, resulting in only short-term fixes or no improvement at all.
This is where hydroxyl radicals, a type of ROS generated by JC-9465, provide a unique advantage. Their high oxidative potential (2.7 eV) allows them to rapidly degrade the molecular bonds in EPS, breaking down the structure from the inside out. This not only eliminates the biofilm but also restores the physical conditions necessary for proper sludge settling.
Case Study: Rosamond CSD Wastewater Treatment Plant
The Rosamond Community Services District, a 2 MGD facility in Southern California, faced severe bulking at the end of summer. Their aerobic digester system—with two 12-foot-deep clarifiers—exhibited a sludge blanket depth of 8–10 feet, indicating major settling issues and threatening compliance limits.
Initial Responses:
- Increased wasting provided temporary relief but was unsustainable.
- Bench testing of cationic flocculants showed increased floc size, but settling remained poor.
- Microscopic analysis revealed no filamentous organisms, only free-swimming ciliates and lagellates.
This scenario reflects a growing trend in wastewater treatment: poor settling even in the absence of filamentous bacteria. Many operators are now discovering that sludge bulking is often driven by organic interference, such as biofilm accumulation and high EPS content—not necessarily microbial type. Unfortunately, traditional detection methods can miss these structural issues, leading to ineffective treatments or misdiagnosed root causes.
Intervention with JC-9465:
After expert consultation, the plant tested JC-9465 at a dosage of 25 mg/L, applied to the RAS line. The impact was swift:
- Within 48 hours, the sludge blanket depth fell to 4–5 feet.
- After 4 days, JC-9465 treatment was stopped.
- The wasting rate was reduced, and operations returned to baseline with no further complications.
Benefits Observed:
- Reduction in wasting rate and overtime associated with plant conditions
- Did not have to take the plant off-line
- Did not require super-chlorination
- Did not require seeding to repopulate the activated sludge population
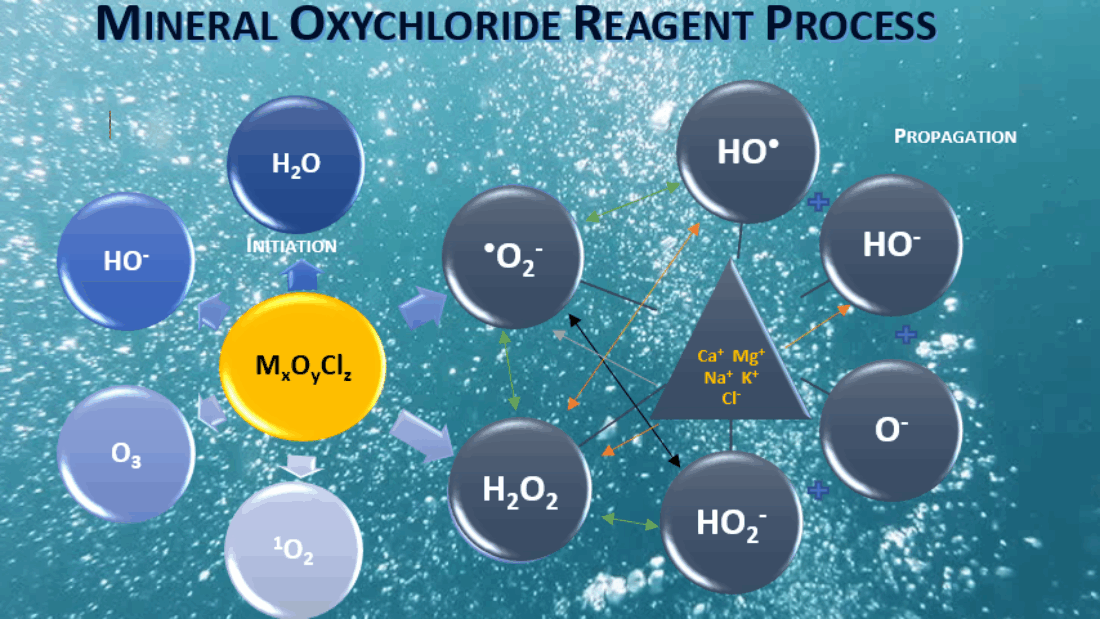
How JC-9465 Works
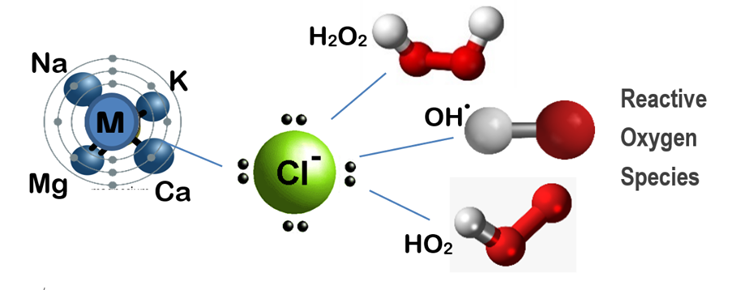
JC-9465 is a mineral oxychloride solution that produces large amounts of hydroxyl radical ions, among the most powerful oxidants used in water treatment. These radicals break molecular bonds within EPS structures, degrading the polysaccharides, lipids, nucleic acids, and proteins that hold the biofilm together.
With an oxidation potential of 2.7 eV, hydroxyl radicals surpass both ozone (2.04 eV) and sodium hypochlorite (1.34 eV) in reactivity. This allows them to initiate fast and irreversible reactions with organic materials, turning complex EPS molecules into simple carbohydrates and dissolved solids. The result is a rapid collapse of biofilm integrity, restoring the natural settling ability of activated sludge.
Sludge bulking is not always the result of filamentous bacterial overgrowth. In many modern wastewater plants, biofilms and EPS accumulation are the hidden causes of poor sludge settling. Traditional treatments may provide temporary relief but often fail to address this core issue.
JC-9465, powered by reactive oxygen species, represents a next-generation solution. It provides fast, targeted action against biofilms without harming essential microbes or requiring aggressive interventions. For operators facing chronic bulking problems, JC-9465 offers an effective, reliable, and process-safe alternative to legacy methods.
If you would like to participate in our research study, please contact us at www.jenfitch.com or email charles@jenfitch.com to discuss the testing protocol and the technology.