Foam is one of the most frustrating challenges in wastewater treatment operations. While it may seem like just a cosmetic issue, uncontrolled foam can have serious consequences: it carries solids into the effluent, disrupts secondary clarification, creates odor problems, and even poses safety hazards for operators working around basins and tanks. Traditional methods such as water sprays, defoamers, or chlorine dosing provide only short-term relief and often leave operators fighting the same problem week after week.
In recent years, operators and engineers have begun to recognize that foam is not just a surface problem. It’s often the result of complex microbial activity and biofilm development within the sludge system. That’s why a chemistry-based approach using Reactive Oxygen Species (ROS), delivered through JC-9465, has gained traction. Unlike surface treatments, JC-9465 directly targets the root causes of foam, making it a much more reliable and long-lasting solution.
Why Foam Happens
Foam usually develops when conditions in the aeration basin favor certain filamentous bacteria like Nocardia or Microthrix parvicella. These organisms thrive under high sludge ages, nutrient imbalances, and elevated grease or surfactant loads. Because of their hydrophobic cell surfaces, they easily trap air bubbles, producing thick, stable foam that resists normal collapse.
Another key contributor is EPS (Extra Polymeric Substances), sticky compounds secreted by bacteria. EPS forms a structural matrix that not only stabilizes biofilms but also makes foam more persistent by binding bubbles together. This is why simple water sprays or even polymers often fail and the foam’s stability is locked into its chemical structure, not just its physical bubbles.
Hidden Costs of Foam
Beyond being unsightly, persistent foam can lead to:
- Effluent quality issues – foam can carry solids over into the clarifier weirs
- Pathogen and odor problems – foam layers can harbor bacteria and create foul smells
- Increased operational effort – constant spraying, chemical dosing, and sludge handling
- Safety concerns – slippery walkways and overflows near open tanks
Recognizing foam as a process symptom rather than just a nuisance is the first step to controlling it effectively.

How JC-9465 Knocks Down Foam
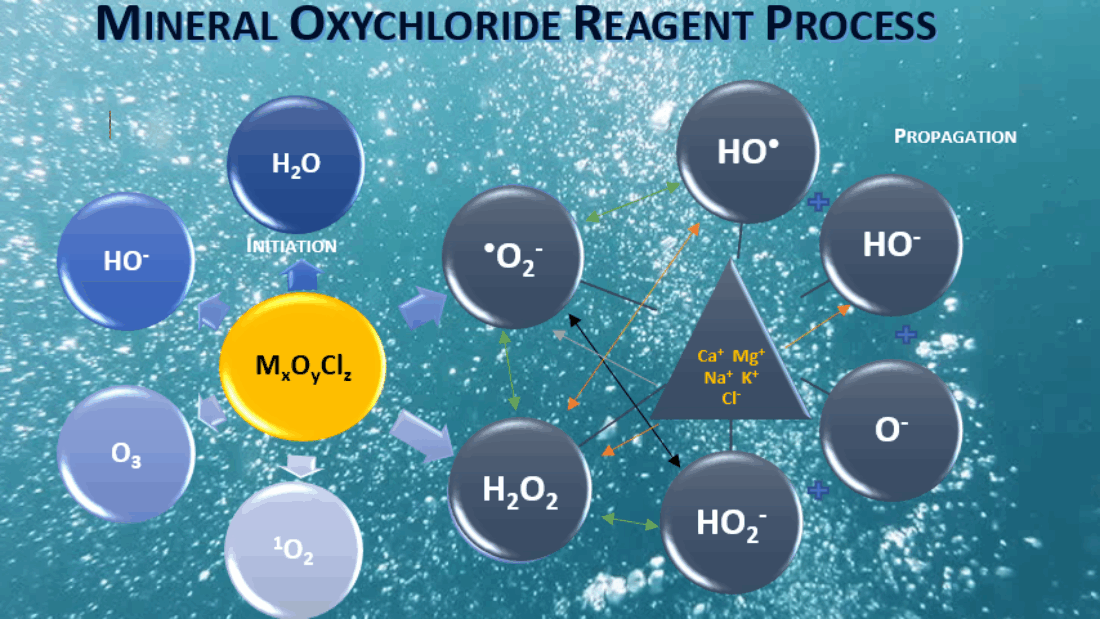
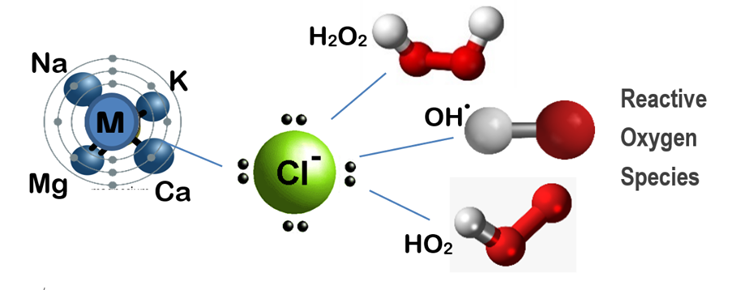
JC-9465 is a mineral oxychloride solution that generates high levels of hydroxyl radical ions, one of the strongest Reactive Oxygen Species (ROS). These radicals have an oxidation potential of 2.7 eV, much higher than ozone (2.04 eV) or sodium hypochlorite (1.34 eV). This high oxidative energy allows JC-9465 to attack and break down the compounds that make foam so stubborn.
The key lies in how JC-9465 interacts with the EPS matrix and foam-causing organisms:
- Disrupting the EPS Matrix
Hydroxyl radicals target and oxidize the compounds that make up EPS, such as proteins, polysaccharides, lipids, and nucleic acids, breaking the ‘glue’ that holds foam together. Once the structural integrity is gone, the foam quickly collapses. - Selective Action on Filamentous Bacteria
Unlike chlorine, JC-9465 selectively targets foam-causing filamentous organisms like Nocardia while preserving beneficial floc-forming microbes, maintaining stable sludge performance. - Residual Oxidative Protection
Beyond immediate knockdown, JC-9465 leaves a residual oxidative effect, helping prevent foam rebound and reducing the need for frequent treatments
Residual Benefits
Another advantage is that JC-9465 doesn’t just provide a quick knockdown; it leaves a residual oxidative effect in the system. This prevents immediate rebound, meaning operators can enjoy longer-lasting control with fewer interventions. In practice, this translates into reduced chemical use, less labor for foam control, and a more stable activated sludge process.
Benefits Compared to Traditional Foam Control
Temporary vs Long-lasting Impacts: Traditional methods of foam control, such as water sprays, defoamers, or chlorination are widely used, but each comes with significant limitations. Water sprays are the simplest and least costly option, but their impact is short-lived. They collapse surface bubbles temporarily, requiring frequent operator attention, and do nothing to address the underlying cause of foam formation.
Solution to Root Cause: Silicone or oil-based defoamers provide a more immediate and dramatic knockdown, but they act only on the surface. Since they don’t penetrate or break down the EPS matrix, they fail to solve the root problem. As a result, foam often returns within hours or days, leading to high recurring chemical costs and dependency on continuous dosing.
Favourable to Beneficial Organisms: Chlorination has long been used to control foaming, especially when filamentous bacteria like Nocardia are identified. While it can reduce microbial populations, chlorination is non-selective and may harm the beneficial floc-forming organisms that are essential for healthy sludge operation. On top of that, chlorine use increases the risk of toxic byproducts such as trihalomethanes (THMs) and chloramines, which can compromise both environmental compliance and safety.
Sustainable Approach: JC-9465, on the other hand, delivers a more effective and sustainable approach. By generating hydroxyl radicals, it directly breaks down the EPS structure that stabilizes foam while selectively reducing foam-causing bacteria. This dual action results in rapid foam knockdown and prevents the rebound that commonly follows conventional methods. Unlike chlorine, it does not disrupt the overall microbial balance, and unlike silicone defoamers, it leaves a residual protective effect that minimizes the need for repeated applications. Over time, this means fewer chemical additions, reduced operator labor, and a more stable biological process.

Case Study: Proven Biofilm and Foam Control with JC-9465
Extensive studies demonstrate the power of JC-9465 in breaking down biofilms and restoring process efficiency. One of the most compelling results comes from the work on controlling Legionella, a pathogen that thrives inside biofilm aggregates.
In these trials, JC-9465 was applied and achieved a 6-log reduction of Legionella in less than 30 seconds. This rapid effectiveness was confirmed through ORP measurements exceeding +680 mV, proving that the hydroxyl radicals generated by JC-9465 were strong enough to penetrate and oxidize the EPS matrix protecting these microbes. Once the EPS was dismantled, the biofilm collapsed and eliminated the safe harbor that allowed Legionella to persist.
The same principle applies in wastewater treatment when controlling persistent foaming issues. Foam, much like biofilm, is stabilized by EPS and the activity of filamentous bacteria. By breaking down EPS and selectively targeting foam-causing organisms, JC-9465 provides fast foam knockdown while preserving the beneficial floc-forming microbes that keep sludge healthy. This is why operators consistently see not only immediate relief, but also longer-lasting stability compared to traditional defoamers or chlorination.
Conclusion
Foam control in wastewater treatment is far more than a surface issue. It’s a process stability challenge rooted in microbial activity and biofilm formation. While traditional approaches such as spraying, defoamers, or chlorine dosing provide short-term relief, they fail to address the underlying causes and often lead to recurring problems, higher costs, and process disruptions.
JC-9465 offers a proven, chemistry-driven solution by generating hydroxyl radicals that break down the EPS matrix and selectively target foam-causing organisms. This not only delivers rapid knockdown but also ensures longer-lasting control without harming beneficial microbes. As demonstrated in real-world applications, JC-9465 allows operators to regain control of their systems, reduce chemical and labor dependency, and maintain consistent effluent quality.
Therefore, sustainable foam control starts with addressing the root cause. With JC-9465, wastewater operators can shift from reactive, temporary fixes to a proactive, reliable solution that supports long-term plant performance and compliance.